Abstract
Background: CD19 CAR-T cell therapy have brough hope to patients with refractory and relapsed (R/R) B cell malignancies. However, the primary result of CAR-T therapy for patients with R/R AML remained modest. One of the main challenges is that current targets for myeloid malignancies are either widely expressed on healthy hemopoietic stem cells. CAR-T cell could cause lasting bone marrow depression. Therefore, to receive a balance, we design a CD33 CAR to recognize AML cells and using NK cells to replace T cells to eliminate tumor cells. The CD33 CAR NK cells have combined the advantage of tumor associated target CD33 and the safety of NK cells.
Methods: 10 qualified subjects with R/R AML aged between 18 and 65 years-old were enrolled and received round(s) of infusion of anti-CD33 CAR NK cells (6×108, 1.2×109 or 1.8×109 cells per round after the precondition with Fludarabine (30mg/m2) and Cytoxan 300-500mg/m2 for 3 days to 5 days, determined by tumor burden at baseline. We investigated the response rate and treatment related side-effect after the CAR NK cell infusion and recorded the long-term efficacy.
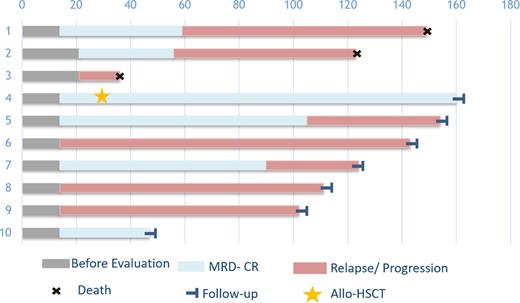
Results: 10 pts have finished efficacy assessment. The median age was 44.5 (range, 18 to 65) years-old and the median treatment lines patients received were 5 lines (range, 3 to 8). The median tumor burden is 20% (range, 8% to 78%). In dose group one, three patients received 3 rounds of CAR NK cells (6×108, 1.2×109 and 1.8×109 cells) with the interval of 7 days. In dose group two, 3 patients received one dose of 1.8 ×109 cells CD33 CAR NK cells. In dose group three, 4 patients received 3 rounds of 1.8 ×109 cells CD33 CAR NK cells with the interval of 7 days. 7(70%) patients developed Grade 1 CRS (cytokine release syndrome), which was alleviated after symptomatic treatment. Only one patient developed grade 2 CRS presented as lasting fever for 6 days after infusion and alleviated after one dose 5mg Dexamethasone I.V. Six of ten patients have received MRD- CR at day 28 assessment. The follow-up was recorded in Figure 1.
Conclusion: Our primary data of the phase I trial have proved the primary efficacy and safety of CD33 CAR NK cells for patients with R/R AML. The efficacy needs expanded samples and longer follow up.
Disclosures
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal